What Is Chondroitin?
Chondroitin is a substance that occurs naturally in the connective tissues of people and animals. As a supplement in higher doses than is found naturally, chondroitin is a popular treatment for osteoarthritis.
Why do people take chondroitin?
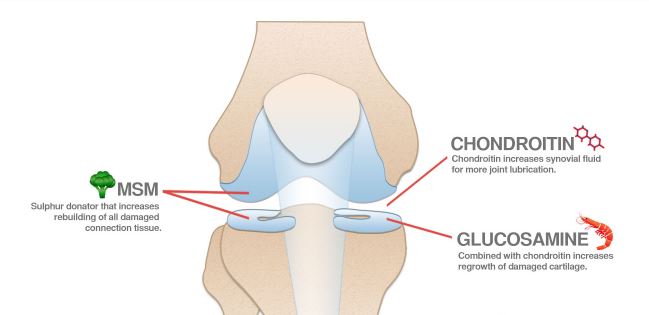
As an osteoarthritis treatment, chondroitin is often used along with another supplement, glucosamine.
The evidence that chondroitin helps with osteoarthritis is mixed. A number of studies seemed to show that it is effective. Researchers found that chondroitin appeared to reduce pain, increase joint mobility, and decrease the need for painkillers.
Chondroitin is a major component of cartilage?
Chondroitin is a major component of cartilage that helps it retain water. It is made by the body naturally. For production of supplements, it can be manufactured from the cartilage of animals, like cows, pigs or sharks, or it can be made in the laboratory. The supplement is sold as chondroitin sulfate. In many European countries it is approved as a prescription treatment for OA. In the U.S., it is often combined with a glucosamine supplement.
Chondroitin sulfate and glucosamine are popular supplements used to treat the pain and loss of function associated with osteoarthritis (OA).
Chondroitin Medical use
Although chondroitin is used in dietary supplements as an alternative medicine to reat osteoarthritis and also approved and regulated as a symptomatic slow-acting drug for this disease (SYSADOA) in Europe and some other countries, it is technically neither a medicine nor a disease-modifying treatment. It is commonly sold together with glucosamine.
Chondroitin Clinical Studies
Some of the most relevant recently conducted clinical trials performed with chondroitin sulfate are summarized below.
- Clinical trials performed to assess the symptomatic effect of chondroitin sulfate in osteoarthritis:
The largest trial conducted with the product is the Glucosamine and Chondroitin Arthritis Intervention Trial (GAIT), a double-blind, randomized, multicenter clinical trial sponsored by the US National Institutes of Health in 1583 patients with knee osteoarthritis, which was published in the New England Journal of Medicine (Clegg DO, et al. 2006). Patients were randomly assigned to one of five orally administered treatments: two 250 mg capsules of glucosamine hydrochloride three times daily, two 200 mg capsules of chondroitin sulphate three times daily, two capsules of 250 mg of glucosamine hydrochloride plus 200 mg of chondroitin sulphate three times daily, 200 mg of celecoxib daily, or placebo. Treatment was administered for 24 weeks.
- Primary Outcome:
The primary outcome measure was a 20% decrease in the WOMAC pain subscale from baseline to week 24. The analysis of the primary outcome data for all patients showed the percentage of responders in each group to be: glucosamine: 64.0%; chondroitin sulphate: 65.4%; glucosamine + chondroitin sulphate: 66.6%; and celecoxib: 70.1% (p=0.008). Thus, despite the considerable effect elicited by all the products, only the celecoxib group reached statistical significance. This has been discussed by the authors and attributed mainly to the unprecedented high response in the placebo group (60.1%) and the relatively mild degree of pain among participants, which may have limited the ability to detect treatment benefits. Indeed, treatment effects were more substantial in patients with moderate-to-severe pain. Analysis of the results for the primary outcome, based on higher baseline pain, showed the percentage of responders in each treatment group to be 54.3% for placebo; 61.4% for chondroitin sulphate; 65.7% for glucosamine hydrochloride; 69.4% for celecoxib; and 79.2% for combined glucosamine and chondroitin sulphate. Thus, in the higher WOMAC pain stratum, only the combination glucosamine + chondroitin group presented significant efficacy (p=0.002).
- Secondary Outcomes:
Secondary efficacy outcome measures were as follows: OMERACT-OARSI response, 50% decrease in WOMAC pain score, WOMAC pain, stiffness and function score, normalized WOMAC score, patient and investigator global evaluations of disease status and response to study medication, evaluation of the index knee for swelling and tenderness, Health Assessment Questionnaire (HAQ) Alternative Disability score and HAQ Pain score, clinical evaluation for adverse reactions and reconciliation of study medications and rescue analgesia use. Analysis of the OMERACT-OARSI measure in all randomized patients revealed statistical significance for both celecoxib (p=0.007) and glucosamine and chondroitin in combination (P=0.02) compared to placebo. Swelling of study joints was significantly less in patients in the chondroitin sulphate (P=0.01) and celecoxib (P=0.03) treatment arms. Differences in the other secondary outcomes were not statistically significant when compared across the 5 treatment arms for all patients. For patients with moderate-to-severe pain at baseline (n=354), a statistically significant difference was observed in favor of the glucosamine and chondroitin combination group versus placebo for the following efficacy outcomes: 20% decrease in WOMAC pain score (0.002); OMERACT-OARSI response (0.001); 50% decrease in WOMAC pain score (0.02); WOMAC pain score (0.009); WOMAC function score (0.008); normalised WOMAC score (0.017); Health Assessment Questionnaire Pain score (0.03).
While the combination therapy of glucosamine and chondroitin sulfate significantly reduced knee pain in the moderate-to-severe pain subgroup, Clegg et al. (2006) state that another trial needs to confirm their results. The authors also note that there was a small number of patients in the moderate-to-severe pain subgroup, which could have limited the study’s power to demonstrate efficacy. It is concerning that glucosamine and chondroitin in this study only showed efficacy in combination. It is possible that when glucosamine and chondroitin interact there is a synergistic effect; however, a synergistic effect has not been proven (Singh, Noorbalochi, MacDonald, & Maxwell, 2016).
A subanalysis of GAIT results based on interaction of Kellgren & Lawrence Grade and Chondroitin Sulfate response relative to placebo was performed by the same study investigators. The results indicated better response for chondroitin sulfate relative to placebo for Kellgren & Lawrence grade 2 compared with grade 3. In general, chondroitin sulfate response within the Kellgren & Lawrence grade 2 group was very similar to that seen for celecoxib. These results suggest that chondroitin sulfate may improve osteoarthritis knee pain in patients with relatively early radiographic disease.
Sawitzke A, et al. 2010 evaluated the efficacy and safety of glucosamine and chondroitin sulfate, alone or in combination, as well as celecoxib and placebo on painful knee osteoarthritis over 2 years as a continuation of the GAIT trial. This was a 24-month, double-blind, placebo-controlled study, enrolling 662 patients with knee osteoarthritis who satisfied radiographic criteria (Kellgren/Lawrence grade 2 or 3 changes and baseline joint space width of at least 2 mm). This subset continued to receive their randomized treatment (glucosamine 500 mg three times daily, chondroitin sulfate 400 mg three times daily, the combination of glucosamine and chondroitin sulfate, celecoxib 200 mg daily, or placebo) over 24 months. The primary outcome was a 20% reduction in pain over 24 months as measured by the Western Ontario and McMaster University Osteoarthritis Index (WOMAC). Secondary outcomes included an Outcome Measures in Rheumatology/Osteoarthritis Research Society International response and change from baseline in WOMAC pain and function. Over 2 years, none of the treatments (not even the positive control celecoxib) achieved a clinically important difference in WOMAC pain or function as compared with placebo. Adverse reactions were similar among treatment groups and serious adverse events were rare for all treatments.
A recent study called the CONCEPT trial (Reginster, Dudler, Blicharski, & Pavelka, 2017) investigated the efficacy of chondroitin sulfate, glucosamine, and celecoxib in the management of symptomatic knee osteoarthritis. A 6 month, double-blind, double-dummy experiment using 604 patients with knee osteoarthritis was conducted. Patients were randomly assigned to one of three experimental groups: 1. One tablet of pharmaceutical grade chondroitin sulfate 800 mg and one placebo capsule of celecoxib 2. One capsule of celecoxib 200 mg and one placebo tablet of chondroitin sulfate 3. One placebo capsule of celecoxib and one placebo tablet of chondroitin sulfate. Patients in all groups were required to take their capsules and tablets once daily in the evening. The CONCEPT trial (Reginster et al., 2017) found that chondroitin sulfate was as effective as celecoxib. The authors stated that an 800 mg/day pharmaceutical-grade tablet of chondroitin sulfate should be “considered first-line treatment in the medical management of knee OA.”
While it is a prescription or over-the-counter drug in 22 countries, chondroitin is regulated in the U.S. as a dietary supplement by the Food and Drug Administration. In Europe, chondroitin sulfate formulations are approved as drugs with evidenced efficacy and safety demonstrated by clinical trials in osteoarthritic patients.
How much chondroitin should you take?
Since chondroitin is not an established treatment, the ideal dosage is uncertain. For osteoarthritis, some studies have used 1,200 milligrams to 1,800 milligrams once a day. Other studies have used smaller doses multiple times a day. Ask your doctor for advice.
Can you get chondroitin naturally from foods?
Chondroitin occurs naturally in animal tissue, especially connective tissues. Gristle on animal bones is high in chondroitin. However, these sources are much lower than the doses provided in chondroitin supplements. Some chondroitin supplements come from animal sources, like shark or beef cartilage.
What are the risks of taking chondroitin?
- Side effects. Chondroitin seems to be safe for most people. Side effects are rare.
- Risks. People who have asthma, or prostate cancer should not take chondroitin supplements without talking to a doctor first. There have been some instances of hyperssensitivity in people with shellfish allergies. Since chondroitin might work as a blood thinner, there is a theoretical risk of bleeding, especially in people with bleeding disorders.
- Interactions. If you take any medications regularly, talk to your doctor before you start using chondroitin supplements. They could interact with drugs like blood thinners and NSAID painkillers or supplements like ginkgo biloba, garlic, and saw palmetto.